Chromatin confomation capture (3C) technologies are used to identify DNA
interactions in the nucleous. These technologies are 3C, 4C, 5C,
ChIA-PET and Hi-C. They all involve crosslinking the DNA in the hopes of
capturing chromatin interaction. Thereafter the DNA is fragmented, often
by restriction enzymes or sonication, forming DNA fragments with free
ends. These free ends can then be ligated, so that the two chromatin
strands that were interacting bind one another, forming only one strand
that can later be sequenced. These strands that form are called chimeric
DNA fragments, as the fragments are formed by fusion of DNA from two or
more loci.
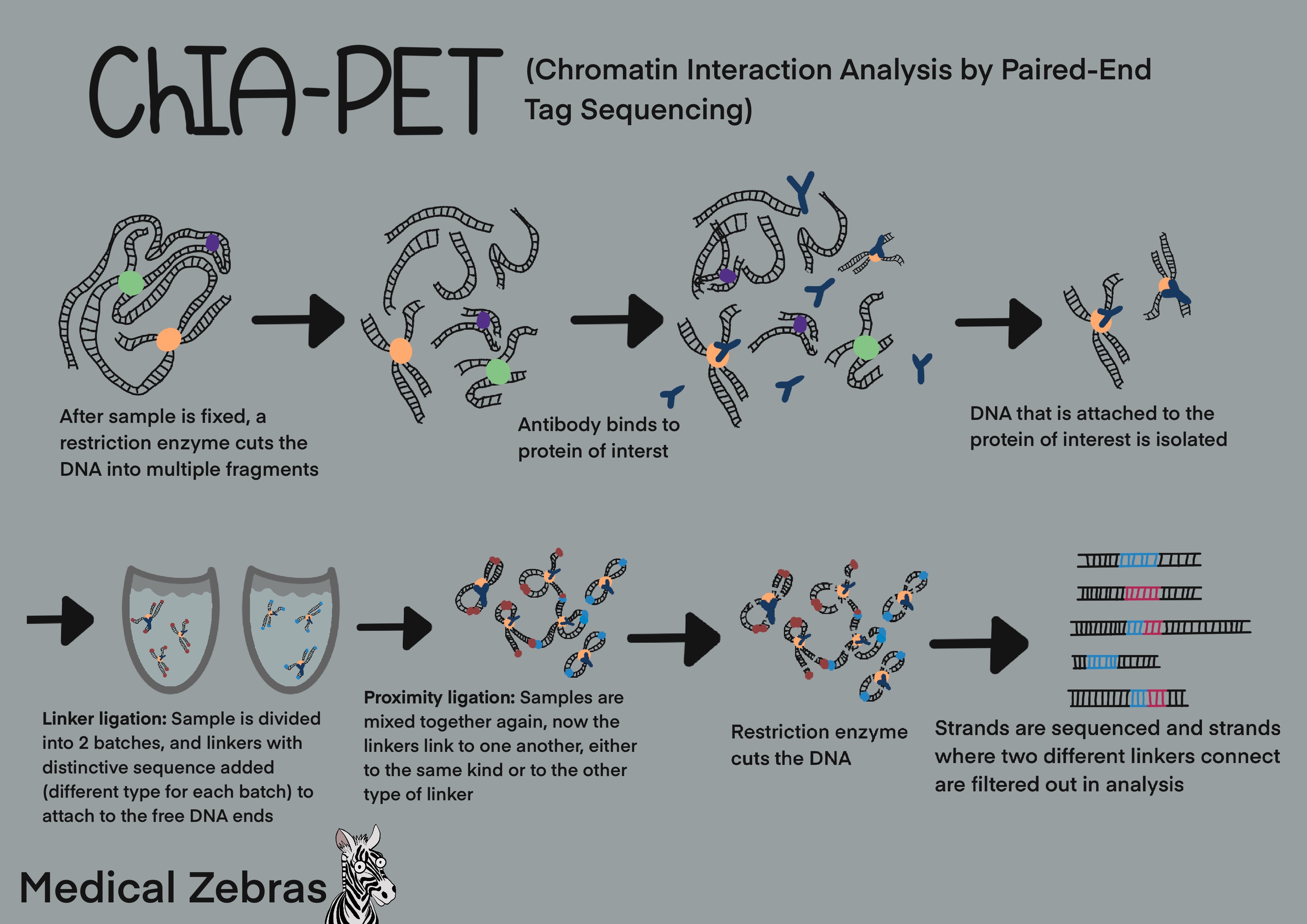
ChIA-PET (Chromatin Interaction Analysis by Paired-End Tag Sequencing)
ChIA-PET is a method developed to better understand the 3-D structure of
the genome and how chromatin interactions play a role in transcription
regulation. This involves first cross-linking the DNA, so that DNA
regions that are in close proximity get cross-linked together, along
with proteins that interact with the DNA. Thereafter the DNA is
fragmented by sonication and immunoprecipitated with an antibody that
attaches to the protein of interest (Antibody that attaches to the
protein of interest and DNA that is attached to that protein is isolated
from the sample). Thereafter the sample is divided into two aliquotes
and one type of linkers are added to each aliquote with specific
sequence that can be identified later during analysis. These linkers
attach to the free ends of the DNA fragments. After mixing the aliquotes
together again, the linkers self-ligate to proximal linkers, forming DNA
loops and thereby capturing what parts of the chromatin have been in
proximity to the protein of interest and other chromatin. The DNA sample
is then cut with a restrictive enzyme so that the DNA loops now form
strands, that can be prepared for further analysis and sequencing. After
sequencing the strands, the strands that contain two linkers of the same
type are identified and the sequences on either side of were the linkers
attached aligned to the genome. Thus identifing which areas of the DNA
have been in close proximity to one another.

Pros: Can be used to detect both long-range and
short-rage chromatin interactions for the protein of interest.
Cons:
Limited sensitivity and large amount of cells needed (>100 million) Examples of uses in the field
~ Not used in the clinical setting.
Cons:
Limited sensitivity and large amount of cells needed (>100 million) Examples of uses in the field
~ Not used in the clinical setting.
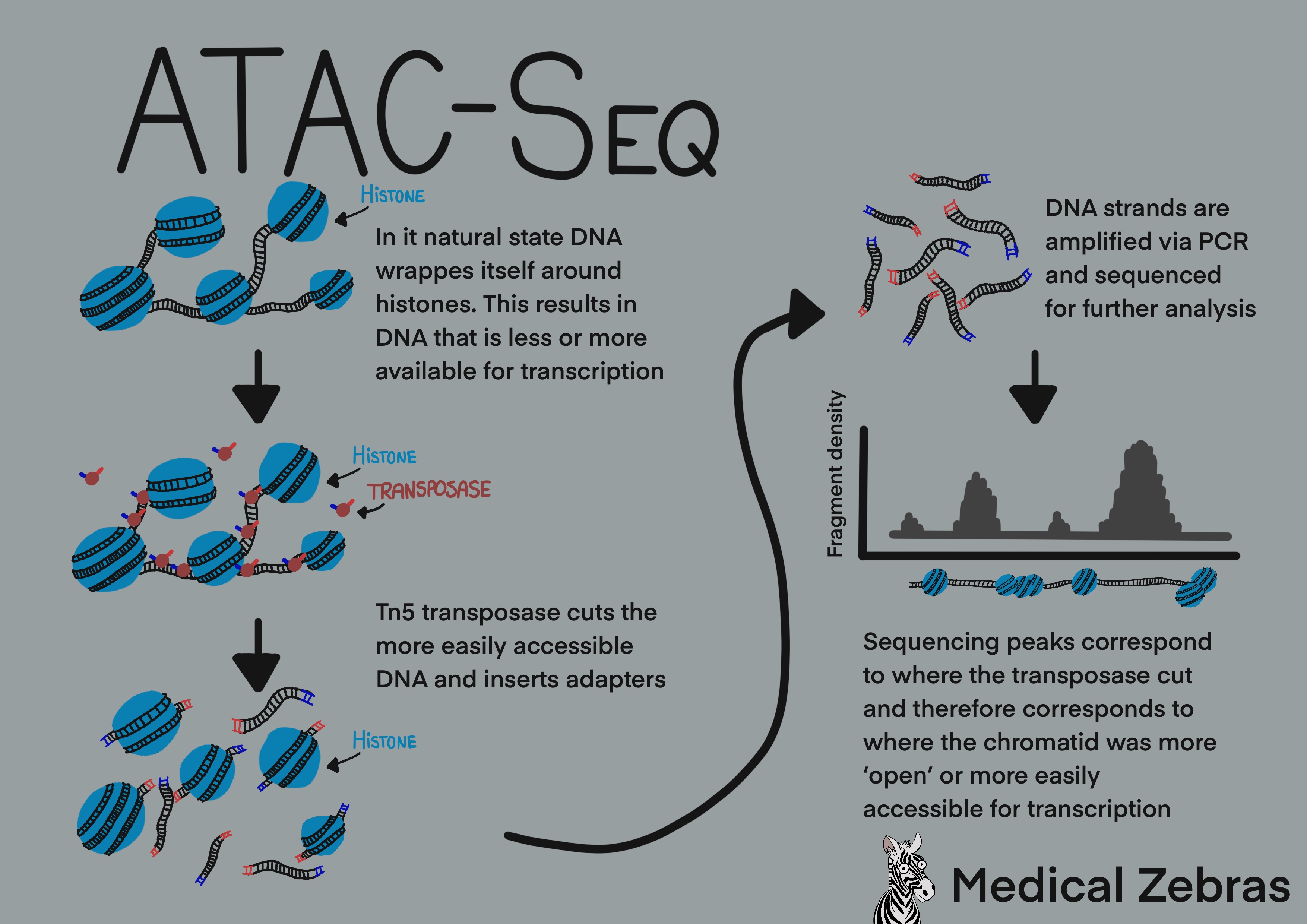
ATAC-Seq
ATAC-seq or Assay for Transposase-Accessible Chromatin using sequencing,
is a method used for investigating chromatin accessibility. A
hyperactive mutant Tn5 transposase is added to a DNA sample. This
transposase cuts double-stranded DNA where it is accessible (not bound
around histones). As well as cutting the DNA the transposase inserts
adaptors to the DNA. These adaptor tagged DNA fragments can then be
isolated, amplified and later sequenced for identification. Regions that
are more often sequenced and identified, correlate to a more open
chromatin. Meaning that the transposase had more frequent access to it
compared to other areas of the genome.
Pros: a relatively fast and simple analysis.
Cons: Tn5 transposase has a sequencing bias that can affect the end results (it does not insert itself into DNA at random).
Examples of uses in the field
~ Not used in the clinical setting.
Cons: Tn5 transposase has a sequencing bias that can affect the end results (it does not insert itself into DNA at random).
Examples of uses in the field
~ Not used in the clinical setting.